|
THE HYDROGEN ECONOMY
Please use our A-Z INDEX to navigate this site where page links may lead to other sites, see HOME
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
CHALLENGES - At present, the biggest challenge for the implementation of this technology in automotive and shipping applications is the complexity of hydrogen storage. Gas hydrogen at normal pressure and temperature conditions has a lot of energy per Kg but this is not the case of its volumetric base. To reduce its volume, it has to be compressed and stored at around 700 bar. To contain the gas at this pressure, the tank needs to have walls so thick that its weight is heavier than the gas inside. In these conditions, the contained energy per tank Kg to hydrogen ratio is drastically lower. Other alternatives for hydrogen storage are the liquid hydrogen tanks (−252,87 °C) and the storage in solid oxides. The first alternative requires high gas liquefaction costs and highly thermal isolating tanks. Also, some metal hydrides can store hydrogen in their crystal lattice and then release it at high temperatures. This technology is very promising although its current efficiency is very low.
The
hydrogen economy is the use of hydrogen as a low carbon fuel, particularly for heating, hydrogen vehicles, seasonal energy storage and long distance transport of energy.
CSIRO - Hydrogen-powered cars also have more distance range – about 800 km compared to between 160-500 km for electric cars. And if the hydrogen is derived from renewable energy sources, hydrogen-powered cars are emissions free.
However costs can be prohibitive to wider uptake, as most retail for about $80,000. Dolan said fuel for these hydrogen-powered cars would cost about $15/kg, with a tank holding about 5 kg of hydrogen.
Two cars powered by ultra-high purity hydrogen derived from ammonia were successfully tested in Brisbane, Australia. This development is the culmination of nearly a decade’s worth of work on membrane technology by CSIRO researchers. The membrane is a modular unit that can be installed at a refuelling station. It derives ultra-high purity hydrogen from ammonia, while blocking all other gases.
Why bother? Why not use battery cars with solar panels attached as an integral part of the bodywork. Australia was home to the Darwin to Adelaide solar car races that were so fast they had to raise the speed limit. With so much sunshine to tap into, it makes little sense complicating electric vehicles, other than constantly seeking more sustainable solutions.
SAFETY
Hydrogen has one of the widest explosive/ignition mix range with air of all the gases with few exceptions such as acetylene, silane, and ethylene oxide. That means that whatever the mix proportion between air and hydrogen, a hydrogen leak will most likely lead to an explosion, not a mere flame, when a flame or spark ignites the mixture.
This makes the use of hydrogen particularly dangerous in enclosed areas such as tunnels or underground parking. Pure
hydrogen-oxygen flames burn in the ultraviolet color range and are nearly invisible to the naked eye, so a flame detector is needed to detect if a hydrogen leak is burning. Hydrogen is odorless and leaks cannot be detected by smell.
The European Commission has funded the first higher educational program in the world in hydrogen safety engineering at the University of Ulster. It could be that the general public will be able to use hydrogen technologies in everyday life with at least the same level of safety and comfort as with today's fossil fuels, but they said that about zeppelins and along came the Hindenburg. We should also remember the number of space craft that have exploded on take off.
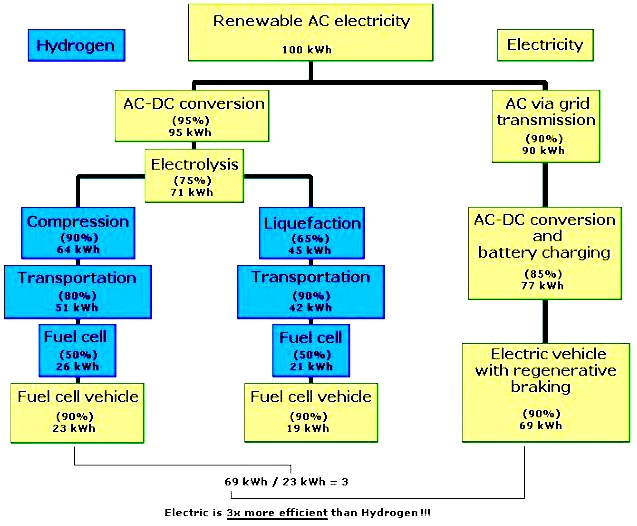
TRANSPORT FUEL EFFICIENCY
The energy that must be utilized per kilogram to produce, transport and deliver hydrogen (i.e., its well-to-tank energy use) is approximately 50 MJ using technology available in 2004. Subtracting this energy from the enthalpy of one kilogram of hydrogen, which is 141 MJ, and dividing by the enthalpy, yields a thermal energy efficiency of roughly 60%.
Gasoline, by comparison, requires less energy input, per gallon, at the refinery, and comparatively little energy is required to transport it and store it owing to its high energy density per gallon at ambient temperatures. Well-to-tank, the supply chain for gasoline is roughly 80% efficient (Wang, 2002). Another grid-based method of supplying hydrogen would be to use electrical to run electrolysers. Roughly 6% of electricity is lost during transmission along power lines, and the process of converting the fossil fuel to electricity in the first place is roughly 33 percent efficient.
Thus if efficiency is the key determinant it would be unlikely hydrogen vehicles would be fueled by such a method, and indeed viewed this way, electric vehicles would appear to be a better choice. However, as noted above, hydrogen can be produced from a number of feedstocks, in centralized or distributed fashion, and these afford more efficient pathways to produce and distribute the fuel.
Although Ford Motor Company and French Renault-Nissan cancelled their hydrogen car R&D efforts in 2008 and 2009, respectively, they signed a 2009 letter of intent with the other manufacturers and Now GMBH in September 2009 supporting the commercial introduction of FCVs by 2015.
A study by The Carbon Trust for the UK Department of Energy and Climate Change suggests that hydrogen technologies have the potential to deliver UK transport with near-zero emissions whilst reducing dependence on imported oil and curtailment of renewable generation. However, the technologies face very difficult challenges, in terms of cost, performance and policy.
DAIMLER-CHRYSLER - A Mercedes bus converted to run on hydrogen from a fuel cell. Hydrogen is simply a method to store and transmit energy. Energy development of various alternative energy transmission and storage scenarios which begin with hydrogen production, but do not use it for all parts of the store and transmission infrastructure, may be more economic, in both near and far term.
An alternative to gaseous hydrogen as an energy carrier is to bond it with nitrogen from the air to produce ammonia, which can be easily liquefied, transported, and used (directly or indirectly) as a clean and renewable fuel. For example, researchers at CSIRO in Australia in 2018 fuelled a Toyota Mirai and Hyundai Nexo with hydrogen separated from ammonia using a membrane technology.
COSTS
The barrier to lowering the price of high purity hydrogen is a cost of more than 35 kWh of electricity used to generate each kilogram of hydrogen gas. Hydrogen produced by steam reformation costs approximately three times the cost of natural gas per unit of energy produced.
This means that if natural gas costs $6/million BTU, then hydrogen will be $18/million BTU. Also, producing hydrogen from electrolysis with electricity at 5 cents/kWh will cost $28/million BTU — about 1.5 times the cost of hydrogen from
natural
gas. Note that the cost of hydrogen production from electricity is a linear function of electricity costs, so electricity at 10 cents/kWh means that hydrogen will cost $56/million BTU. However costs are rapidly decreasing.
Hydrogen accelerates the cracking of
steel (hydrogen embrittlement), which increases maintenance costs, leakage rates, and material costs. The difference in cost is likely to expand with newer technology: wires suspended in air can use higher voltage with only marginally increased material costs, but higher pressure pipes require proportionally more material.
PILOT PROGRAMS
United States has a hydrogen policy with several
examples. A joint venture between NREL and Xcel Energy is combining wind power and hydrogen power in the same way in Colorado. Hydro in Newfoundland and Labrador are converting the current wind-diesel Power System on the remote island of Ramea into a Wind-Hydrogen Hybrid Power Systems facility. A similar pilot project on Stuart Island uses solar power, instead of wind power, to generate electricity. When excess electricity is available after the batteries are fully charged, hydrogen is generated by electrolysis and stored for later production of electricity by fuel cell.
Western Australia's Department of Planning and Infrastructure operated three Daimler Chrysler Citaro fuel cell buses as part of its Sustainable Transport Energy for Perth Fuel Cells Bus Trial in Perth. The buses were operated by Path Transit on regular Transperth public bus routes. The trial began in September 2004 and concluded in September 2007. The buses' fuel cells used a proton exchange membrane system and were supplied with raw hydrogen from a BP refinery in Kwinana, south of Perth. The hydrogen was a byproduct of the refinery's industrial process. The buses were refueled at a station in the northern Perth suburb of Malaga.
UTOPIAN
The allure of the hydrogen economy is plain, splitting plain ordinary water using electrolysis to obtain oxygen and hydrogen gas is like a dream come true, especially if we can generate free electricity using solar cells and wind turbines to split the water.
Then the hydrogen is free right. But is the electricity free?
No, not really.
There is a cost, including the cost of manufacturing the solar panels or wind turbines and the transmission line installation and maintenance.
Where there is a cost, then we have to consider payback time and working life. If we can use most of the solar and wind energy directly to power vehicles, we make the best of the working life of our energy harvesting apparatus. And that means reduced greenhouse gases, so a reduced carbon footprint for the human race in an anthropogenic fight against climate change.
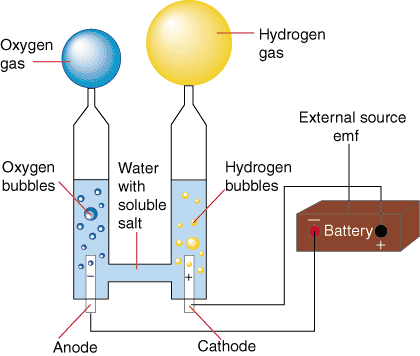
BACK TO BASICS - ELECTROLYSIS
A water molecule is formed by two elements: two positive Hydrogen ions and one negative Oxygen ion.
The water molecule is held together by the
electromagnetic attraction between these ions. When
electricity is introduced to
water through two electrodes, a cathode (negative) and an anode (positive), these ions are attracted to the opposite charged electrode. Therefore the positively charged hydrogen ions will collect on the cathode and the negatively charged oxygen will collect on the anode.
EUREKA
-
Hydrogen is the most abundant element in the universe. With the "green-energy" craze and talk of powering our future oil-free economy on hydrogen, it has received much attention in the last few decades. Learning about this potential fuel of the future is important and
interesting, but not without snags, and these are for anyone to seek to
overcome.
ANIMATION
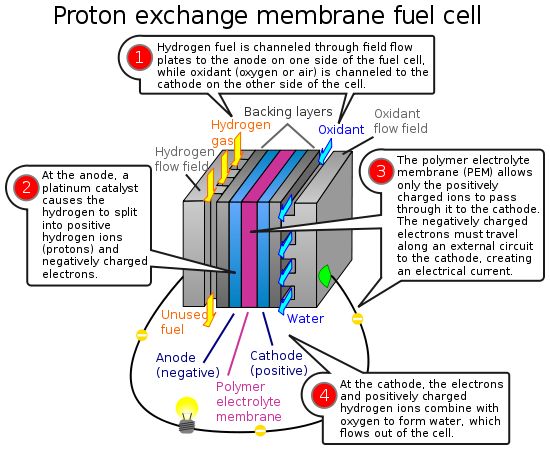
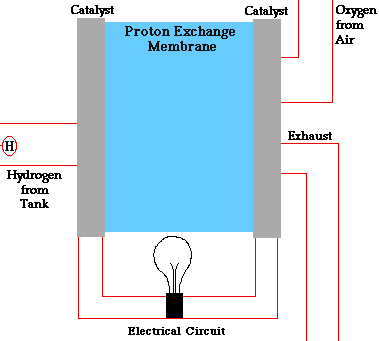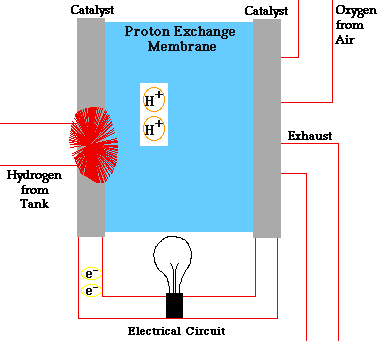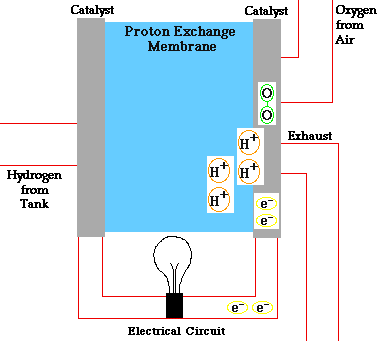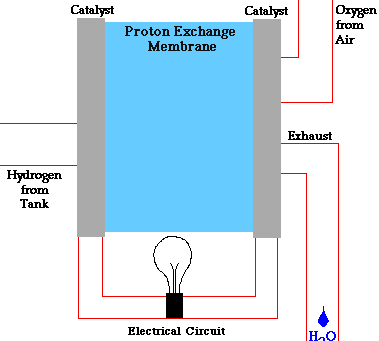
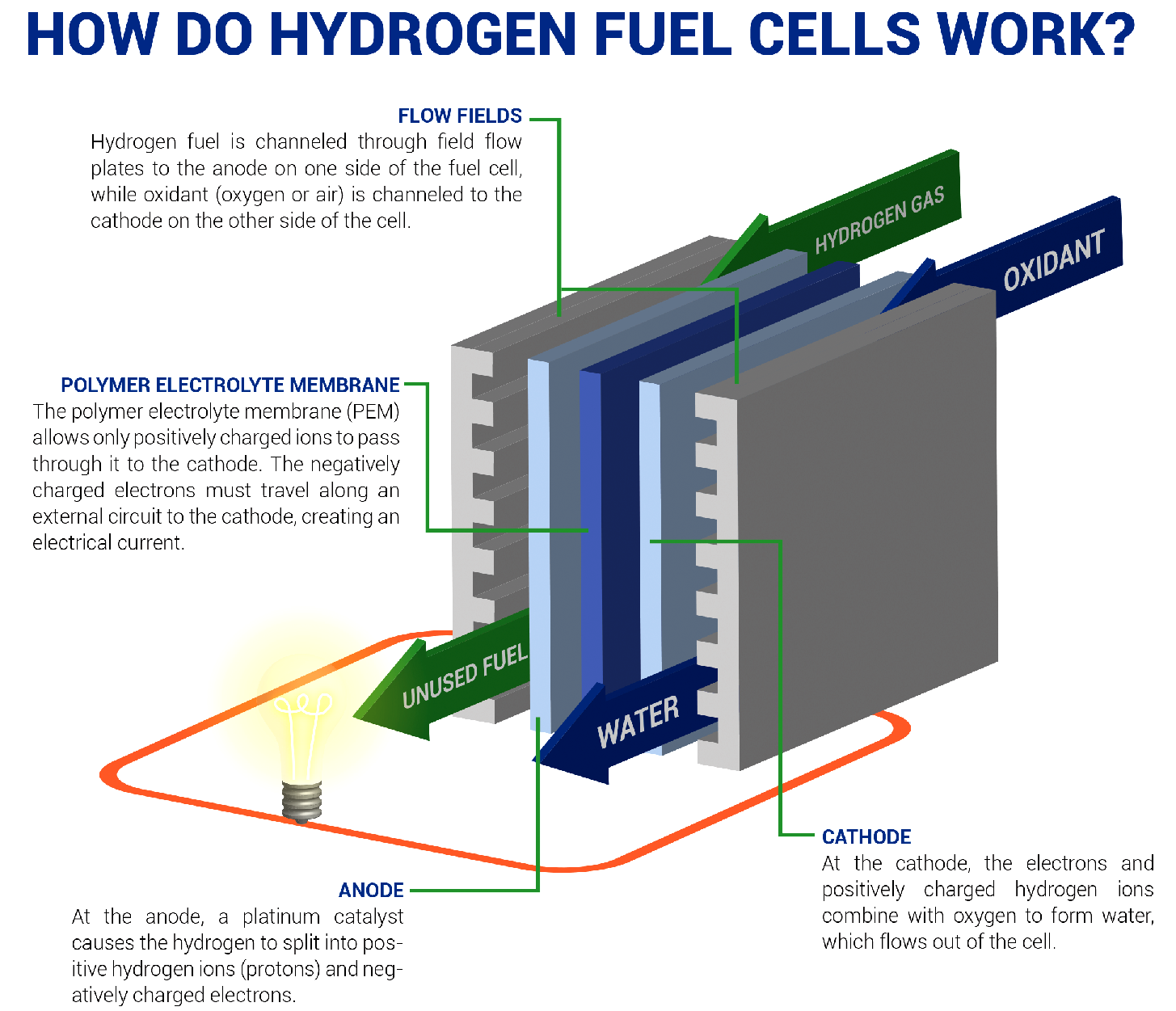
- A fuel cell converts the chemicals hydrogen and oxygen into water, and in the process it produces electricity.
A-Z
INDEX OF H2 POWERED FUEL CELL SHIPS
A - Z INDEX OF SPONSORS AND ASSOCIATES - WORLD HYDROGEN CONGRESS
LINKS & REFERENCES
https://www.theengineer.co.uk/hydrogen-economy/ https://edu.rsc.org/feature/fuelling-the-future-solid-phase-hydrogen-storage/2020153.article https://www.cnbc.com/2019/02/21/musk-calls-hydrogen-fuel-cells-stupid-but-tech-may-threaten-tesla.html https://www.cnbc.com/2019/02/26/how-toyota-is-helping-japan-create-a-hydrogen-fueled-society.html https://www.ft.com/content/98080634-a1d6-11e7-8d56-98a09be71849 https://www.explainthatstuff.com/fuelcells.html
...
USA - In 2003, President Bush announced a program called the Hydrogen Fuel Initiative (HFI) during his State of the Union Address. This initiative, supported by legislation in the Energy Policy Act of 2005 (EPACT 2005) and the Advanced Energy Initiative of 2006, aims to develop hydrogen, fuel cell and infrastructure technologies to make fuel-cell vehicles practical and cost-effective by 2020. Obviously, the legislation did not work, or we'd seen hydrogen cars selling like hot cakes. Whereas, there are significant sales of battery electric cars.
The United States has dedicated more than two billion dollars to fuel cell research and development so far. Yet the basics principles of climate change is to find the best way to use less energy to achieve the same goal. Of course we have to explore all avenues before deciding on what works best. Thomas Edison found 1,000 ways not to make a light bulb before inventing his carbon filament version that succeeded. Joseph Swan in the UK filed a similar patent before the more famous US inventor. Keep at it chaps.
Please use our A-Z INDEX to navigate this site
AMMONIA - COMPRESSED GAS - ECONOMY - FUEL CELLS - FUSION - HYDRIDES - LIQUID GAS - METHANOL
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
This website is provided on a free basis as a public information service. copyright © Climate Change Trust 2022. Solar Studios, BN271RF, United Kingdom.
|